Carbon Monoxide: Deadly killer on the loose
Carbon monoxide, the stubborn stalker
A month ago, the public in the Netherlands was shocked by the news that a 27-year-old male student from The Hague was arrested in a triple homicide. Police said he attacked and killed innocent pedestrians in public parks with a knife.
The student had an earlier history of suspected psychic troubles and that used anti-depressants. But his fellow students and neighbors considered him as a quiet and intelligent person who kept to himself and who would not harm anyone.

However, this shocking triple homicide fits a pattern of deadly domestic and public violence by psychiatric patients who use antidepressants. The past three to four years there were at least 15 such cases in the Netherlands alone, with a remarkably large proportion being committed by women
In the US there are similar cases of extreme violence by psychiatric patients – the ‘Prozac killings’ -, and every time a public discussion erupts about the question if anti-depressants have side effects that promote or stimulate violent behavior. This explanation seems to be accepted more or less in the USA, as a reason for reduced responsibility. But Dutch courts of law do not accept such excuses and the judges usually hand out stern verdicts for deadly ‘psychic’ violence.
However, lately, there were signals that indicate another possibility for these mysterious cases of extreme violence, namely intoxication by carbon monoxide, in combination with antidepressants or other psychoactive drugs.
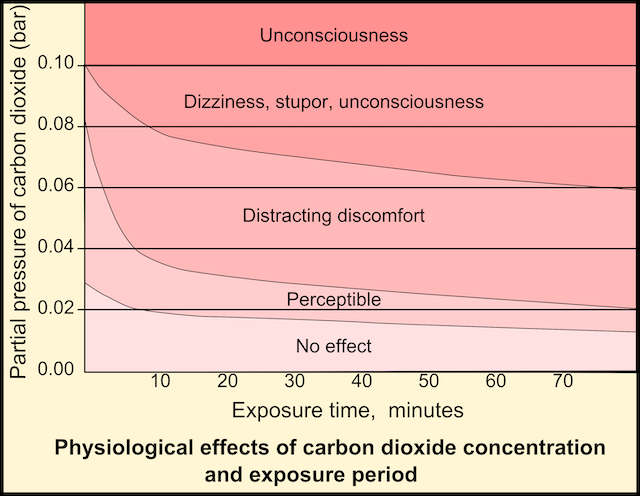
In 2016 an Amsterdam policeman who used two anti-depressants entered a home that was intoxicated by carbon monoxide from a gas-fired water heater. Once inside the house, the policeman got an acute panic attack and killed the occupant with four shots, although witnesses later declared that they saw no dangerous situation.
The 27-year-old student from The Hague, had recently moved into a room where he quickly became psychotic and violent, authorities said. This room was located above the kitchen of a large restaurant in an old building; a suspect situation for carbon monoxide.
In 2008 the ‘Netherlands Journal for Psychiatry’ published an article ‘Chronic carbon monoxide intoxication: a psychotic depression brought on by a new kitchen’[i]. This article was about the dangers of carbon monoxide in kitchens, but it also suggested that psychiatric patients should be tested for carbon monoxide: “If psychiatrists are better informed about the affective and neuropsychological symptoms that frequently accompany carbon monoxide poisoning, they should be able to recognize the symptoms sooner.”
This is a true but neglected advise, unfortunately, because nowhere in the world psychiatric patients are routinely tested for carbon monoxide intoxication. Also in this tragic recent case of the triple homicide, neither the offender nor his room were immediately tested for carbon monoxide; a simple test that might have explained his sudden outburst of blind rage. However, in such cases the forensic investigators should act fast to collect evidence, because in the environment carbon monoxide (CO) is easily converted into the harmless carbon dioxide (CO2), while the half-time of carbon monoxide in human blood is only four hours, so within a day, the evidence will be gone.
The WHO and the CDC consider carbon monoxide as an important health issue. This is fully justified, given the abundant presence of carbon monoxide in all energy consuming societies. Wherever carbon-based fuels are burned, carbon monoxide can be found, which explains why carbon monoxide intoxication is the most common intoxication, all over the world.
The website of the CDC www.cdc.gov gives much useful and practical information about carbon monoxide: where it can be found; where the dangers are and how these can be avoided.
The WHO asks special attention for bad side-effects when carbon monoxide and drugs are mixed: “There is some evidence that interactions between drug effects and carbon monoxide exposure can occur in both directions; that is, carbon monoxide toxicity may be enhanced by drug use, and the toxic or other effects of drugs may be altered by carbon monoxide exposure. […] Because of the effect of carbon monoxide on brain function, interactions between carbon monoxide and psychoactive drugs could be anticipated”(WHO. Environmental Health Criteria 213, Carbon Monoxide; par. 1.7.2).
The WHO concludes that not enough research has been done in this field, but signals from society, like the ‘Prozac killings’, are reason enough for grave concern. This WHO warning about the effect of carbon monoxide on brain function and the possible negative interactions between carbon monoxide and drugs or medication should convince medical doctors and psychiatrists that patients should always be checked for carbon monoxide intoxication before any medication is given.
The same applies to those patients where medication does not help (20% of the cases) or where medication makes the situation even worse (5%). Carbon monoxide is a neurotoxin, so it is plausible that in cases of carbon monoxide intoxication medication does not help, or makes matters even worse.
So what is this carbon monoxide?
Carbon monoxide is composed of one atom carbon and one atom oxygen, chemical formula CO. It is a colorless, tasteless and odorless gas that is a by-product of the combustion of carbon-based fuels. It is highly toxic and can cause a wide spectrum of physical and psychic problems and diseases, from mild to fatal within seconds, depending on the concentration. Since the symptoms of CO intoxication are similar to those of many other diseases, misdiagnosis commonly occurs, and a suspected case of CO intoxication must always be confirmed by a chemical test of the blood or breath.
Besides the fact that our senses cannot detect the poisonous CO, there are several other reasons why it is so extremely dangerous:
- Our blood loves CO. The oxygen-carrying hemoglobin in our blood has a 250 fold preference for CO over oxygen, which means that only 1% CO in the air is enough to cause coma in 10 seconds and death within 10 minutes. When the CO blocks the hemoglobin, the internal organs will suffer from lack of oxygen, the most sensitive ones first: brain, heart, and lungs. This is an ‘acute’ form of CO intoxication.
- Apart from being an efficient oxygen inhibitor, CO is also a neurotoxin that will affect important functions of the brain and nervous system. The symptoms are many and varied, depending on the place where the CO attacks the nervous system from within. This will gradually happen at low concentrations, so it is a ‘chronic’ form of CO intoxication. However, there is no clear distinction between acute and chronic intoxication. Acute intoxication may also cause later symptoms that are similar to chronic illnesses, like depression or chronic fatigue syndrome.
- The CO molecule is tiny and invasive, being 12.5% smaller and than a molecule of oxygen. Wherever oxygen goes inside the body or environment, CO arrives there just as easy! This mobility makes CO especially risky for pregnant women because it passes the placenta and may damage the nervous system of the unborn child.
- This may come as a surprise, but the human body itself also produces small amounts of carbon monoxide, about 10 ml per day that is exhaled through the lungs. A healthy non-smoker who lives in a clean environment exhales about 2-4 ppm in his breath. This ‘endogenous CO’ (as opposed to ‘exogenous CO’ from external sources) is a subject of medical speculation and research because it might have a function against infections.
Because CO is so extremely toxic, the concentration is usually expressed in ppm (parts per million), where 10.000 ppm is equal to 1%. It is generally accepted that below 10 ppm there is no danger for human health and that above 20 ppm it is necessary to take action. The WHO advises that 25 ppm is a concentration that is acceptable for a maximum of one hour.
 Common symptoms of ‘acute’ CO intoxication are:
Common symptoms of ‘acute’ CO intoxication are:
- Flu-like symptoms, headache, throat ache, ‘blocked’ head.
- Lack of breath, chest pain, heart palpitations.
- Nausea, vomiting.
- Sensory delusions, hallucination, confusion.
- Weakness, sleepiness, coma, death.
Some symptoms of chronic CO intoxication are:
- Burn-out, depression, chronic fatigue syndrome (CVS), psychoses.
- Agitation, nervousness, ‘short fuse’, aggression and fury.
- Loss of memory and concentration, behavioral problems.
- Incontinence, loss of balance, unsteady walk.
- Bizarre neurologic symptoms, change in personality.
- Anxiety and panic attacks, self-mutilation, suicidal attempts.
- Prolonged CO intoxication may lead to an asymmetric, ‘tell-tale’ face and loss of dentition. [ii]
The websites of WHO and CDC supply much information about the dangers of CO and ways to avoid these. The most important one is being aware of the risks of CO in the environment and knowledge about the most dangerous sources of CO, like combustion engines without catalytic converter (generator sets, lawnmowers, chainsaws, outboard engines, airplanes); heaters and cooking equipment on wood, charcoal, natural gas, propane or kerosene; barbecues and campfires. Using such fuels in confined spaces with limited ventilation, like tents, campers, and boats greatly increase the risk of intoxication.
In most countries, trucks and passenger cars are equipped with a catalytic converter in the exhaust system, that converts CO into the harmless CO2. But this is not a 100% secure solution, because a cracked manifold or leaking gasket may still cause serious problems. Also, an idling engine, when driving very slowly or standing in queues, is a risk, because idling engines always produce more CO, while at the same time the temperature of the catalytic converter may fall below the minimum operating temperature.
Expensive cars sometimes have an alarm that warns when to shut the fresh air supply, so that you do not inhale the poisonous exhaust fumes of the cars in front of you. Having a CO alarm in the car is a good advice for people who often stand in queues (but shutting off the fresh air for too long, will increase the CO2 concentration inside the car, which will make you sleepy and dull!) When shutting off the fresh air supply, one may also try to open a side window, because on busy roads the most toxic air is close to the ground.
Airplanes have no catalytic converter, so there is a risk in single-engine airplanes, where the pilot and passengers sit directly behind the engine and the exhaust system. The FAA sees CO as a deadly menace for such airplanes and advises that they should always have a CO alarm in the cockpit.[iii]
But also large commercial jet airplanes have risks, and there have been several incidents with CO intoxication of passengers and crew. Such incidents may be caused by leaking seals or gaskets in the jet-engines because in most airplanes the fresh cabin air is bled from the engine compressors.
Another risk is congested airports, where planes have to queue and wait in line with idling engines before they may take off. All that time they are surrounded by the exhaust fumes of the planes before them, which will inevitably enter the cockpit and passenger cabin. An idling jet engine has a much higher CO concentration in the exhaust gasses than when running at operational altitude and speed. And strangely enough, commercial airplanes are not required to have CO alarms in the cockpit and pantries.
The question is if CO is an aspect of the mysterious ‘airsickness syndrome’ that affects crewmembers and frequent flyers because the symptoms of CO intoxication and air sickness syndrome are very similar.
It also raises the question if CO intoxication can be a cause of crashes and other serious incidents with airplanes because CO is known to impair mental abilities and sound judgment, also of pilots. Any crash investigation – like the recent crashes of two Boeing 737’s – should include thorough research into a possible CO intoxication of pilots and passengers. Also in the case of the mysteriously disappeared Malaysian Airlines Flight 370 on March 3, 2014, the possibility of a fatal CO intoxication of the crew and passengers immediately comes to mind.
A still very common source of CO is smoking. It is therefore not strange that smokers are more susceptible to symptoms like depression and burn-out. Especially dangerous are water pipes because they use burning charcoal to keep the process going. This means that a water pipe smoker may inhale up to 30 times more CO than a smoker of cigarettes or cigars. And since CO hardly dissolves in water, the water filter does not help at all to prevent intoxication.[iv]
Fortunately, protection against the dangers of CO is not difficult or expensive. The most important factor is awareness, always and everywhere. And there are simple CO alarms for sale for $ 20 or more at many shops and on the internet. However, one should know that these alarms are usually programmed for the higher concentrations (above 100 ppm) that cause acute CO intoxication. It is advisable to purchase such an alarm with a digital read-out for 10 ppm and higher, so even if the alarm does not sound, one can see that something is wrong. More sensitive alarms, also with an adjustable alarm level and visible and audible alarm signals, are available but more expensive.
An interesting protection for people who work in high-risk areas are small data-loggers for CO, that can be carried in a breast pocket. These can store thousands of measurements during a given period, and be read out in any computer with a USB connection. [v]
It is weird to think that carbon monoxide has been a daily companion of the human race for at least as long as we know and use fire. Our bodies have a high affinity for it, they even produce some carbon monoxide and it may have unknown functions that keep us healthy. And yet our bodies never have grown immune or even accustomed to it. It can kill us in seconds without feeling anything or knowing what happened. And after having done such fatal work, it disappears into nothingness without leaving a trace.
Carbon monoxide is probably the most fascinating and common compound in the human environment, and yet so little is known about it.
[i]See http://www.tijdschriftvoorpsychiatrie.nl/issues/260/articles/1744and https://www.ncbi.nlm.nih.gov/pubmed?term=%22Tijdschr%20Psychiatr%22[Journal]%20AND%2050[Volume]%20AND%204[Issue]%20AND%20235[Pagination]
[ii]http://mcsrr.org/graphics/poeposter.jpg
[iii]https://www.faa.gov/pilots/safety/pilotsafetybrochures/media/CObroforweb.pdf
[iv]https://www.rivm.nl/en/tobacco/harmful-substances-in-tobacco-smoke/koolmonoxide-co
[v]https://www.lascarelectronics.com/data-loggers/carbon-monoxide/?type=usb
René van Slooten is a leading ‘Poe researcher’, who theorizes that Poe’s final treatise, ‘Eureka’, a response to the philosophical and religious questions of his time, was a forerunner to Einstein’s theory of relativity. He was born in 1944 in The Netherlands. He studied chemical engineering and science history and worked in the food industry in Europe, Africa and Asia.The past years he works in the production of bio-fuels from organic waste materials, especially in developing countries. His interest in Edgar Allan Poe’s ‘Eureka’ started in 1982, when he found an antiquarian edition and read the scientific and philosophical ideas that were unheard of in 1848. He became a member of the international ‘Edgar Allan Poe Studies Association’ and his first article about ‘Eureka’ appeared in 1986 in a major Dutch magazine. Since then he published numerous articles, essays and letters on Poe and ‘Eureka’ in Dutch magazines and newspapers, but also in the international magazines ‘Nature’, ‘NewScientist’ and TIME. He published the first Dutch ‘Eureka’ translation (2003) and presented two papers on ‘Eureka’ at the international Poe conferences in Baltimore (2002) and Philadelphia (2010). His main interest in ‘Eureka’ is its history and acceptance in Europe and its influence on philosophy and science during the late 19th and early 20th centuries.
